 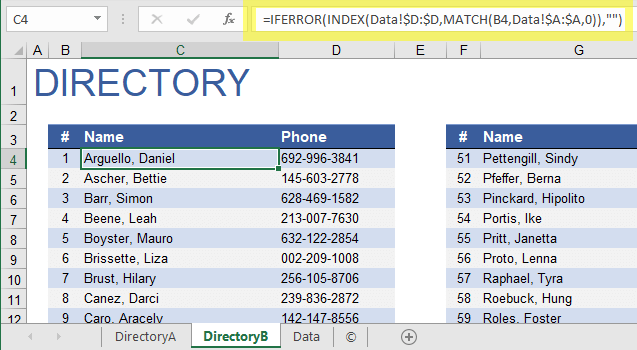
GMP certificates, non-compliance statements and manufacturing authorisations. 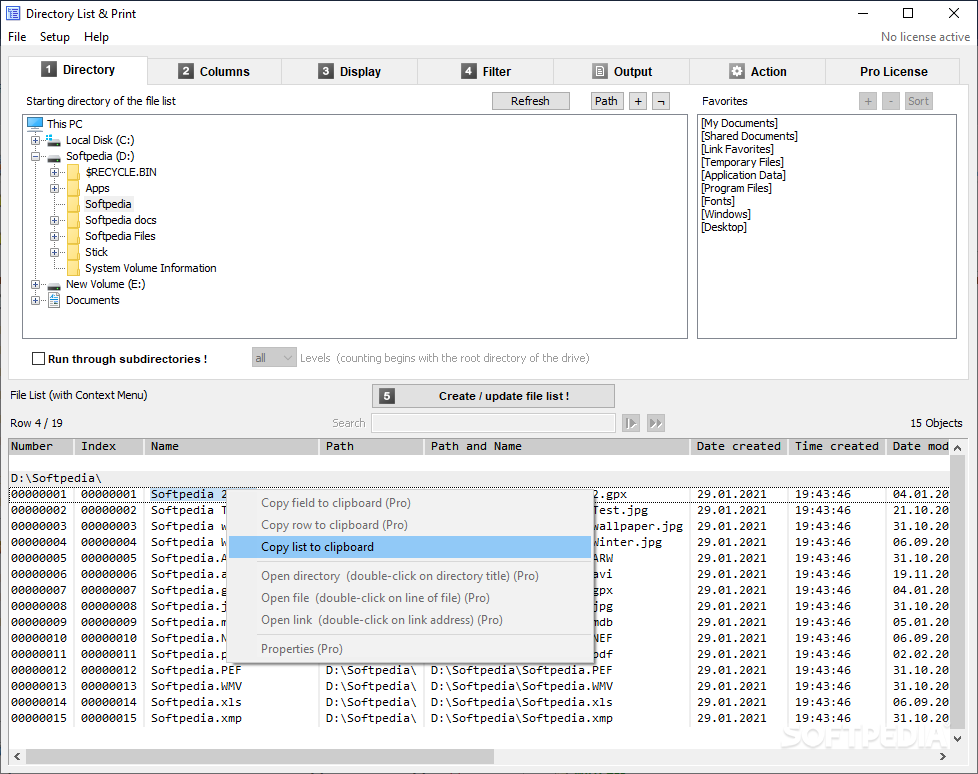
EU GMP guide annexes: Supplementary requirements: Annex 19: Reference and retention samples.Question and answer on residency of the qualified person(QP) - NEW July 2023.Questions and answers on remote batch certification / confirmation by the qualified person (QP) - NEW July 2023.EU GMP guide annexes: Supplementary requirements: Annex 16.EU GMP guide annexes: Supplementary requirements: Annex 13.EU GMP guide annexes: Supplementary requirements: Annex 11: Computerised systems.EU GMP guide annexes: Supplementary requirements: Annex 8: Sampling of starting and packaging materials: Use of near-infrared (NIR) technology for container-wise identity testing.EU GMP guide annexes: Supplementary requirements: Annex 8: Sampling of starting and packaging materials: Glycerol.EU GMP guide annexes: Supplementary requirements: Annex 6: Manufacture of medicinal gases.EU GMP guide annexes: Supplementary requirements: Annex 1: Manufacture of sterile medicinal products.EU GMP guide part II: Basic requirements for active substances used as starting materials: GMP compliance for active substances in investigational medicinal products (IMPs).EU GMP guide part II: Basic requirements for active substances used as starting materials: GMP compliance for active substances.EU GMP guide part I: Basic requirements for medicinal products: Chapter 8: Complaints, Quality Defects and Product Recalls.EU GMP guide part I: Basic requirements for medicinal products: Chapter 7: Outsourced activities.EU GMP guide part I: Basic requirements for medicinal products: Chapter 5: Production.EU GMP guide part I: Basic requirements for medicinal products: Chapter 3: Shared manufacturing facilities.EU GMP guide part I: Basic requirements for medicinal products: Chapter 3: Equipment.EU GMP guide part I: Basic requirements for medicinal products: Chapter 1: Pharmaceutical quality system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
